 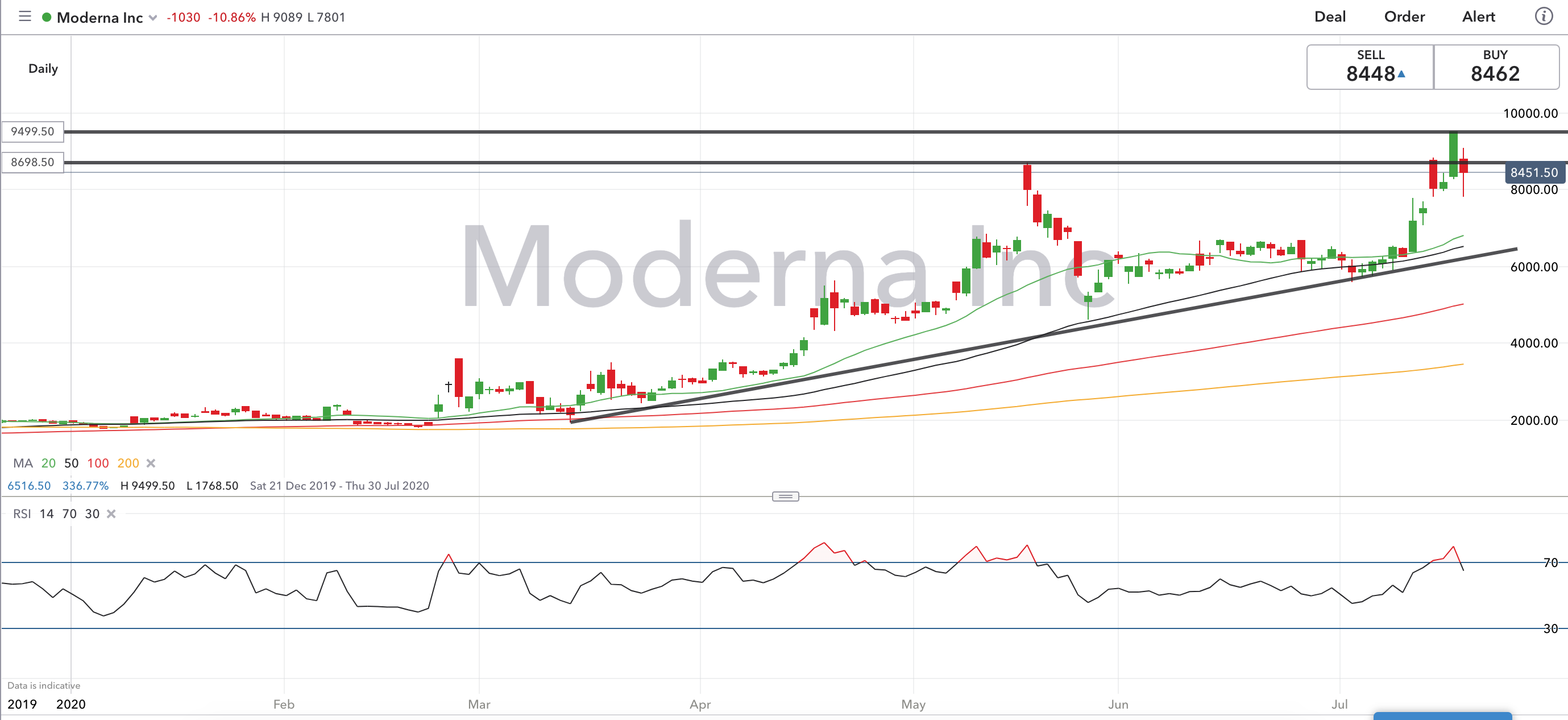
In the judgment of some scientific and medical experts, Moderna’s announcement was premature at best. But many months of further tests involving potentially tens of thousands of human subjects will be needed before the vaccine can be deemed safe and effective against COVID-19 infection and approved by the Food and Drug Administration for wide distribution. The trial was aimed chiefly at determining whether Moderna’s test vaccine caused troublesome side effects the company said that only minor side effects were observed among the initial 45 subjects. Moderna’s vaccine announcement amid the coronavirus outbreak turbocharged its share offering - is there a connection? No raw data were disclosed, nor have any results been published following peer-review thus far.īusiness Column: Moderna’s vaccine results boosted its share offering - and it’s hardly a coincidence The full results from only eight trial subjects were disclosed in the announcement, with full results from 37 other subjects withheld. We reported on the very day of the vaccine announcement that it was based on exceedingly thin data. That probably reflects investor concern about the influx of new shares, as well as doubts expressed in the scientific community about the significance of the vaccine trial results. The shares have since fallen back to less than $62, below its May 15 close. But it was a 5% discount to the stock’s price at the May 18 closing. As we noted at the time, that share price would have been a premium of nearly 14% over the company’s $66.69 price on May 15, the last trading day before the vaccine announcement. Moderna instantly took advantage of the spike by announcing the sale of 17.6 million new shares at $76 each. That announcement on Monday, May 18, produced an immediate spike in Moderna’s stock price, which rose by nearly 20% to close at $80 that day after peaking at $87. “If you sell while you’re there, that doesn’t show a lot of confidence in what you’re doing.” Indeed, several investment advisory services examine buying and selling by corporate insiders for clues about what insiders really think about a company’s prospects.Īnother issue is what the executives’ sales might imply about the timing of the company announcement about its vaccine trial. “Traditionally, when you leave a company, then you sell,” he told me. The company's pipeline also includes a cell therapy-based treatment: a relaxin fusion protein being developed to treat acute decompensated heart failure.Elson is wary of any stock sales by executives during their tenures. Candidates include possible vaccines for influenza, HIV, respiratory syncytial virus, Epstein–Barr virus, the Nipah virus, chikungunya, human metapneumovirus, varicella zoster virus, as well as a cytomegalovirus vaccine, a Zika virus vaccine funded by the Biomedical Advanced Research and Development Authority, and three cancer vaccines. 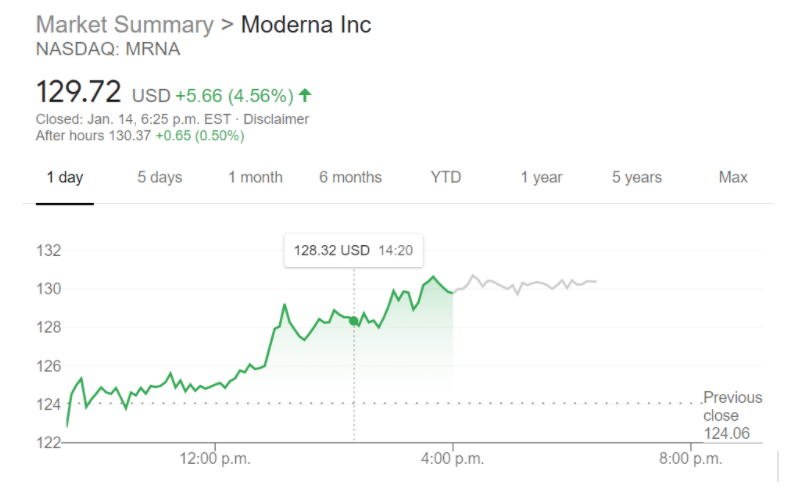
The company has 45 treatment and vaccine candidates, of which 38 have entered clinical trials. The company's only commercial product is the Moderna COVID-19 vaccine, marketed as Spikevax. The company's name is derived from the terms "modified", "RNA", and "modern". These vaccines use a copy of a molecule called messenger RNA to carry instructions for proteins to produce an immune response. 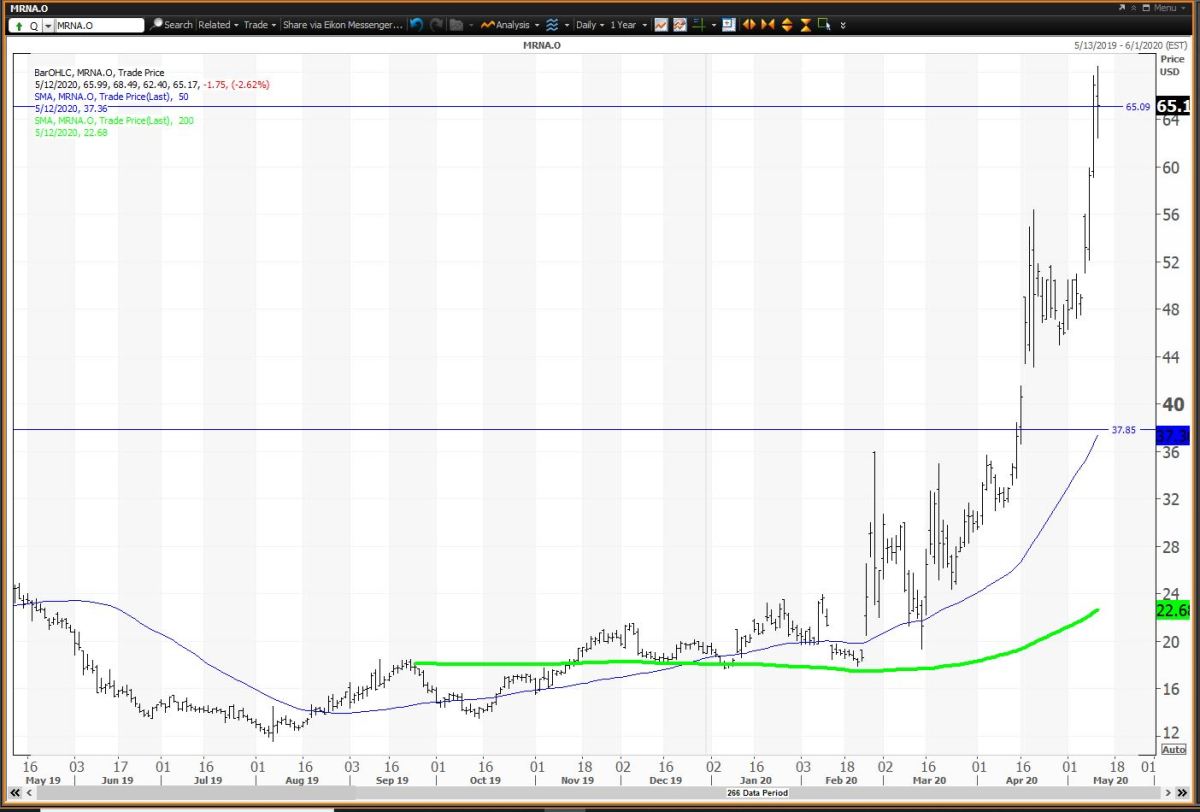
is a pharmaceutical and biotechnology company based in Cambridge, Massachusetts, that focuses on RNA therapeutics, primarily mRNA vaccines.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
